Waec Chemistry Past Questions
Question 1451
Consider the following table| Substitute | Meltig point / ºC | Boiling point / ºC |
| P | - 78 | -25 |
| Q | - 8 | 40 |
| R | - 6 | 150 |
| S | 44 | 280 |
- A. P
- B. Q
- C. R
- D. S
Question 1452
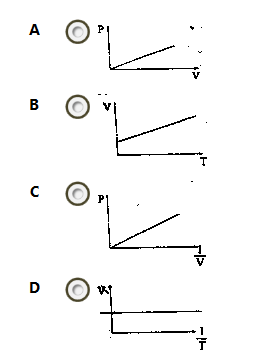
Which of the following sketches is a graphical representation of Boyle's law?
- A. A
- B. B
- C. C
- D. D
Question 1453
Which of the following scientists formulated the law of conservation of mass?
- A. A. Lavoisier
- B. J. Dalton
- C. R. Boyle
- D. J. Proust
Question 1454
The first definition of an element was made by
- A. J. Dalton.
- B. A. Lavoisier.
- C. R. Boyle.
- D. J. J. Thompson.
Question 1455
A reaction is represented by the equation below. A2(g) + B2(g) 2AB(g); ∆H = +X kJmol-1 Which of the following statements about the system is correct?
- A. The reaction goes to completion at equilibrium
- B. The forward reaction is exthermic
- C. At equilibrium increase in temperature favours the reverse reaction
- D. Pressure has no effect on the equilibrium mixture